  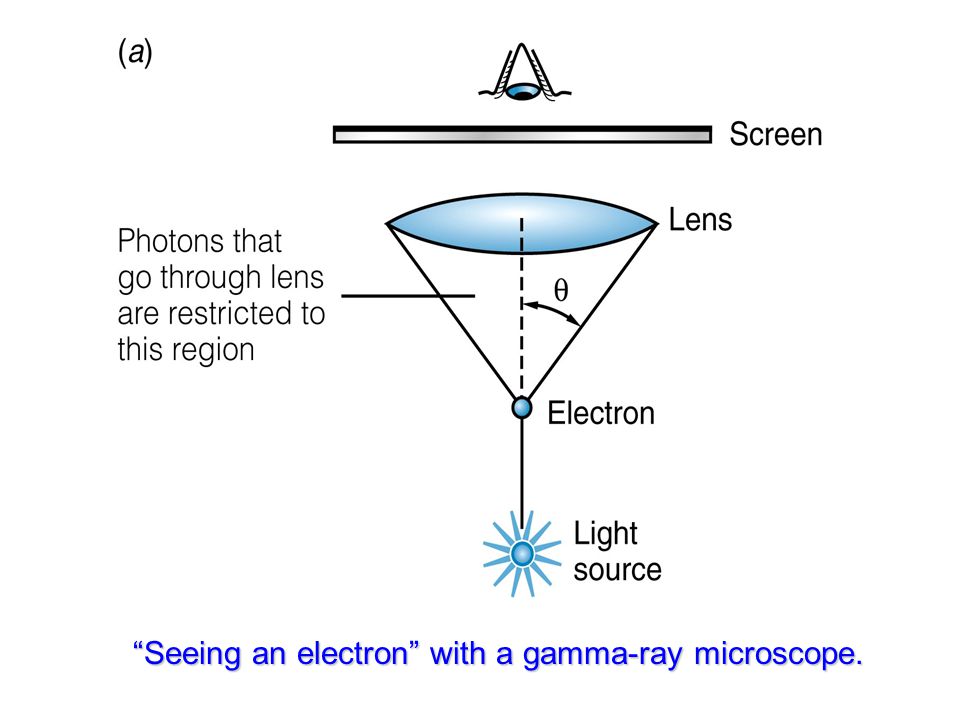
Simply put, the Uncertainty Principle states that the more accurately you know the momentum or energy of a particle, the less accurately you will be able to know it's location in space or time. Heisenberg's Uncertainty Principle on the other hand relates to one of the key concepts of quantum mechanics- wave particle duality. The Pauli Exclusion Principle simply states that no two fermions (a family of particles with half-integer spin, which includes electrons) may exist in the same energy state.Ī good example of this can be seen in the organization of the electrons of an atom into different "shells", each with it's own energy level, where no two electrons have the same energy and momentum.įor a graphic example of this phenomenon, see this website. Consider the double-slit patterns obtained for electrons and photons in Figure 29.25.What is the basic difference between the Pauli Exclusion Principle and the Heisenberg Uncertainty Principle? Let us explore what happens if we try to follow a particle. It is somewhat disquieting to think that you cannot predict exactly where an individual particle will go, or even follow it to its destination. Those who developed quantum mechanics devised equations that predicted the probability distribution in various circumstances. There is a certain probability of finding the particle at a given location, and the overall pattern is called a probability distribution. After compiling enough data, you get a distribution related to the particle’s wavelength and diffraction pattern. However, each particle goes to a definite place (as illustrated in Figure 29.24). The idea quickly emerged that, because of its wave character, a particle’s trajectory and destination cannot be precisely predicted for each particle individually.  
Both patterns are probability distributions in the sense that they are built up by individual particles traversing the apparatus, the paths of which are not individually predictable.Īfter de Broglie proposed the wave nature of matter, many physicists, including Schrödinger and Heisenberg, explored the consequences. (See Figure 29.24.)įigure 29.25 Double-slit interference for electrons (a) and photons (b) is identical for equal wavelengths and equal slit separations. Repeated measurements will display a statistical distribution of locations that appears wavelike. But if you set up exactly the same situation and measure it again, you will find the electron in a different location, often far outside any experimental uncertainty in your measurement. Experiments show that you will find the electron at some definite location, unlike a wave. What is the position of a particle, such as an electron? Is it at the center of the wave? The answer lies in how you measure the position of an electron. Matter and photons are waves, implying they are spread out over some distance. 7.C.1.1 The student is able to use a graphical wave function representation of a particle to predict qualitatively the probability of finding a particle in a specific spatial region.The information presented in this section supports the following AP® learning objectives and science practices: Explain the implications of Heisenberg’s uncertainty principle for measurements.Use both versions of Heisenberg’s uncertainty principle in calculations.By the end of this section, you will be able to: 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
